Meningitis outbreak: Regulators dropped the ball
Sunday, November 18, 2012
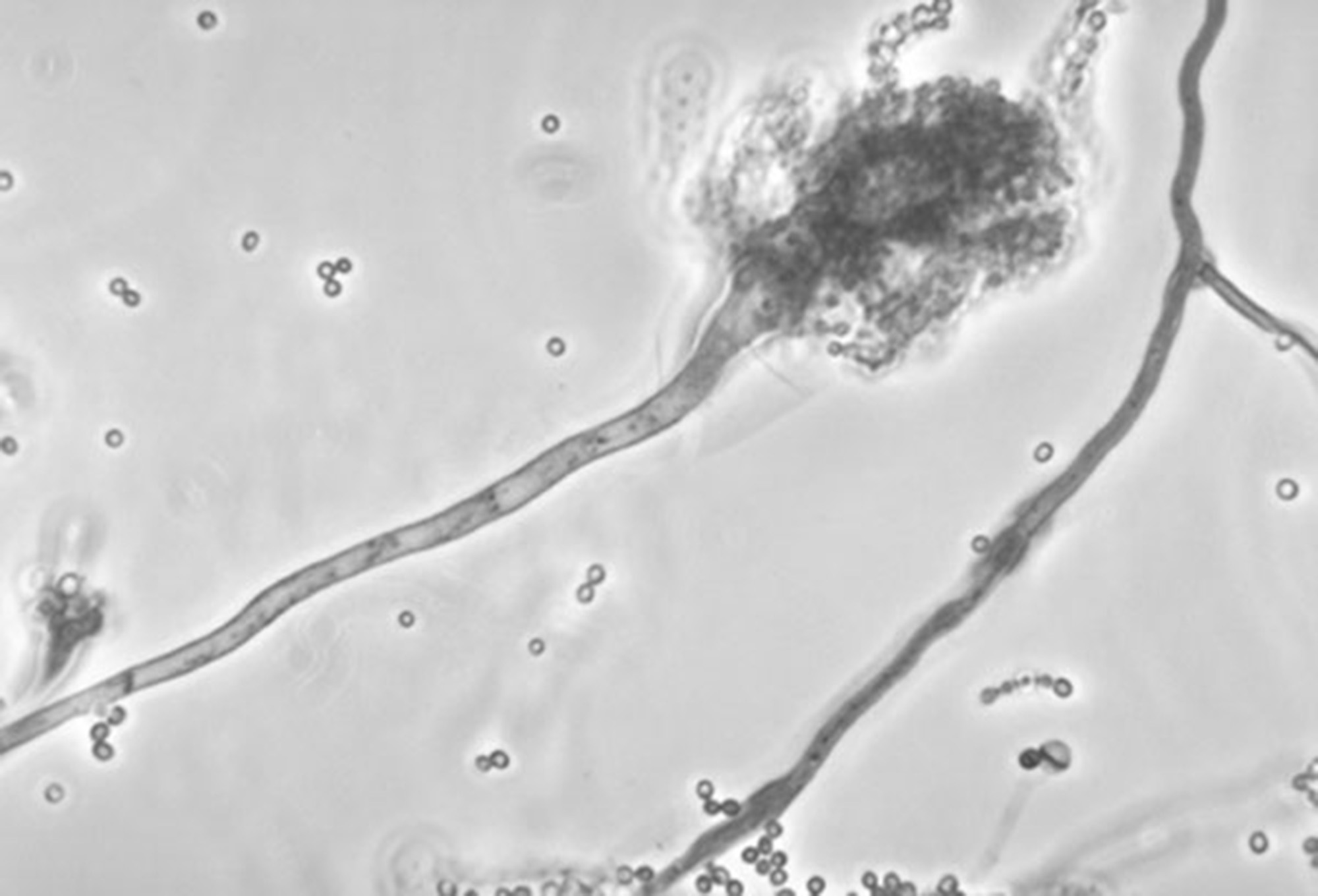 This undated photo made available by the Centers for Disease Control and Prevention shows a branch of the fungus Aspergillus fumigatus. The fungus can also cause skin infections if it enters a break in the skin. The meningitis outbreak is linked to the fungus being accidentally injected into people as a contaminant in steroid treatments. It's not clear how the fungus got into the medicine.
This undated photo made available by the Centers for Disease Control and Prevention shows a branch of the fungus Aspergillus fumigatus. The fungus can also cause skin infections if it enters a break in the skin. The meningitis outbreak is linked to the fungus being accidentally injected into people as a contaminant in steroid treatments. It's not clear how the fungus got into the medicine.On May 10, 2011, long before the current deadly meningitis outbreak, an official in the FDA regional office in Denver sent an email to a colleague in Boston detailing allegations that a Massachusetts drug compounder was illegally shipping drugs to Colorado hospitals.
The information came from Colorado state inspectors who had then passed the concerns along to the U.S. Food and Drug Administration.
But there the information stopped.